Abstract
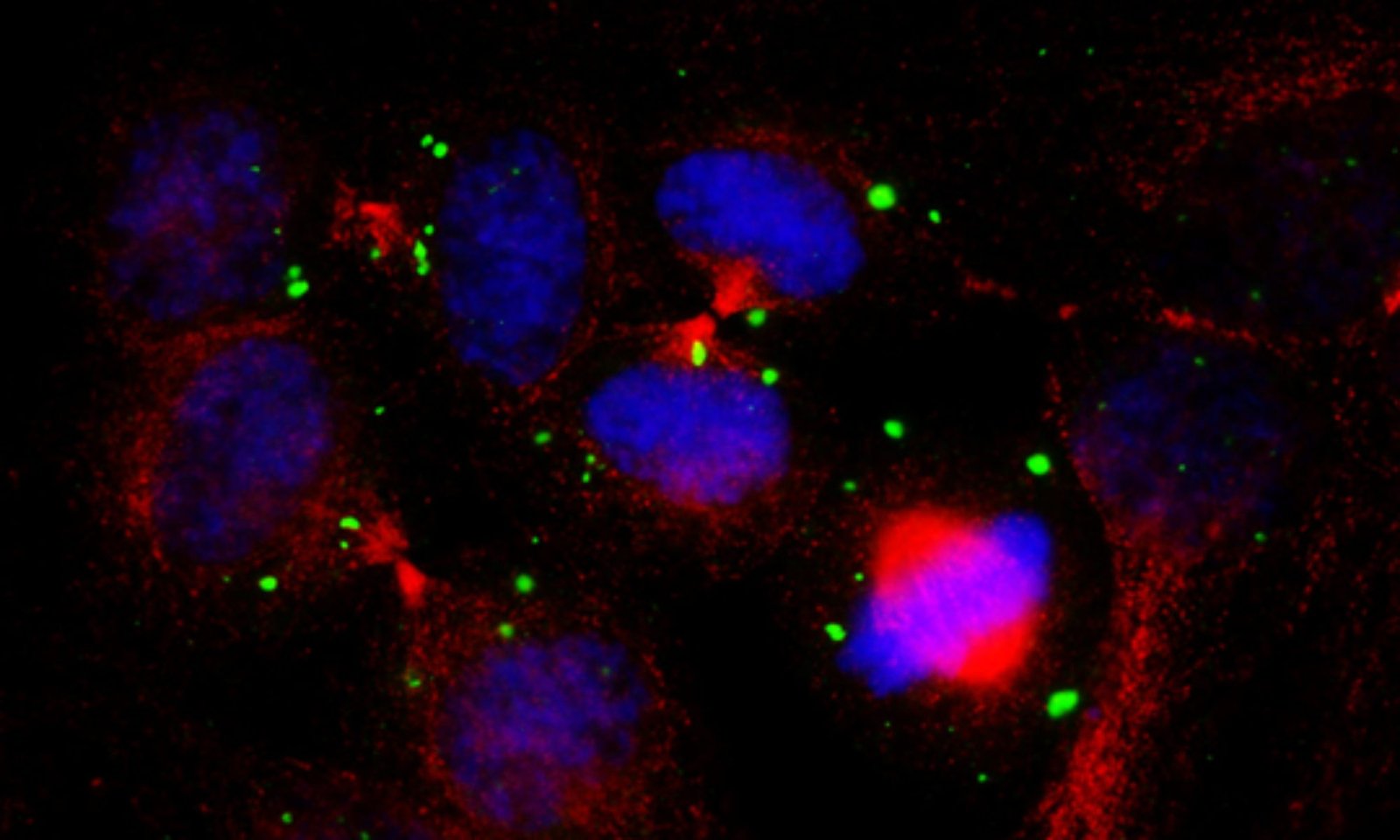
Antimicrobial resistance is a public health emergency and warrants coordinated global efforts. Challenge is that no alternative molecular platform has been identified for discovery of abundant antimicrobial hit compounds. Xanthene libraries have been screened for bioactive compounds. However, the potentially accessible chemistry space of xanthene dyes is limited by the existing xanthene synthesis. Herein we report a mild one-step synthesis, which permits late-stage introduction of a xanthene moiety onto i.e. natural products, pharmaceuticals, and bioactive compounds and construction of a focused library of rhodamine dyes exhibiting facile functional, topographical and stereochemical diversity. In vitro screening yields 37 analogs with mid-to-high bactericidal activity against WHO priority drug-resistant pathogens. These findings suggest that synthetic dye libraries exhibiting high structural diversity is a feasible chemical space combating antibacterial resistance, to complement the natural sources.
Reference:
Luo X, Qian L, Xiao Y, Tang Y, Zhao Y, Wang X, Gu L, Lei Z, Bao J, Wu J, He T, Hu F, Zheng J, Li H, Zhu W, Shao L, Dong X, Chen D, Qian X, Yang Y. A diversity-oriented rhodamine library for wide-spectrum bactericidal agents with low inducible resistance against resistant pathogens. Nat Commun. 2019 Jan 16;10(1):258. doi: 10.1038/s41467-018-08241-3. PubMed PMID: 30651565; PubMed Central PMCID: PMC6335415.
Keywords: diversity-oriented rhodamine library, wide-spectrum bactericidal agents, resistant pathogens, synthetic dye libraries, WHO priority drug-resistant pathogens, xanthene libraries, #AntibioticResistance, #Chemistry.
The International Natural Product Sciences Taskforce (INPST) maintains up-to-date lists with conferences, grants and funding opportunities, jobs and open positions, and journal special issues with relevance for the area of phytochemistry and food chemistry, pharmacology, pharmacognosy research, and natural product science.